
This course provides an exploration of software validation principles within the context of medical device manufacturing.
Duration: 16 hours
CEUs: 1.7
Modality: Group
Participant limit: 15

Test Method Validation (TMV) is a key element to quality and process controls. According to the Title 21 of the Code of Federal Regulations Part 820.72…” Each manufacturer shall ensure that all inspection, measuring, and test equipment, including mechanical, automated, or electronic inspection and test equipment, is suitable for its intended purposes and it is capable of producing valid results.”
Choosing the correct methods can help the organization to minimize risk which could be associated to over-rejection or worst, shipping defective product to a customer.
Learn about the correct methods validation that can help the organization to minimize risk which could be associated to over-rejection or worst shipping product that is defective to customer.
Duration: 12 hours
CEUs: 1.3
Modality: Group
Participant limit: 15
Only logged in customers who have purchased this product may leave a review.
Learn about the correct methods validation that can help the organization to minimize risk which could be associated to over-rejection or worst shipping product that is defective to customer.
Duration: 12 hours
CEUs: 1.3
Modality: Group
Participant limit: 15

This course provides an exploration of software validation principles within the context of medical device manufacturing.
Duration: 16 hours
CEUs: 1.7
Modality: Group
Participant limit: 15
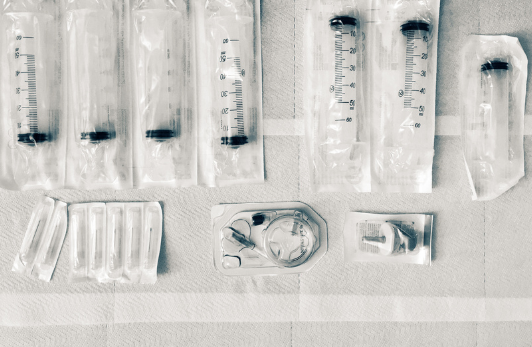
Packaging Fundamentals provides an overview of essential concepts related to packaging in the context of medical devices. It covers the applicable standards and regulatory requirements, an exploration of packaging materials and key topics.
Duration: 16 hours
CEUs: 1.7
Modality: Group
Participant limit: 15

Learn about the 8D Methodology for Root Cause Analysis / Structured Problem Solving.
Duration: 8 hours
CEUs: 0.9
Modality: Group
Participant limit: 15

Learn the entire process validation cycle and the key elements such as VMP, EQ, OQ and PQ, and the correlation with the regulations and standards
Duration: 24 hours
CEUs: 2.5
Modality: Group
Participant limit: 15

There are no reviews yet.